Help to transform our extraordinary hospital into something even better.
- Refine search:
Sisters Ammarah, Summayah and Ayaana are regular visitors to the Clinical Research Facility at Sheffield Children’s – because all three are taking part in a clinical study!
Ammarah, the youngest sister at seven years old, has severe atopic dermatitis which is more commonly known as eczema. This is a condition which causes the skin to become itchy, dry, cracked and sore.
Along with her sisters, Ammarah is taking part in the PELISTAD trial. Run by Sanofi and Regeneron, this study is investigating the effect of the drug dupilumab on the skin barrier function for children aged 6 to 11 years old who have moderate to severe atopic dermatitis. Dupilumab has been approved to use in the UK for patients aged six and older with moderate to severe atopic dermatitis since December 2021.
Ammarah has suffered with eczema since she was five months old. Her mum Safia said: “Her symptoms were so severe that she had never slept a full night in her own bed, instead having to stay with my husband and I because she was in so much discomfort.”
During the study Ammarah had monthly injections of dupilumab while the Research team at Sheffield Children’s monitored her response to the drug. Safia said: “It has made a huge difference to Ammarah’s symptoms – she’s become a much happier child. Since receiving the drug she has been able to sleep in her own bed every night.
“Ammarah was teased at school because of her eczema – her classmates didn’t really understand what it was. Being on the trial has given her the confidence to teach them about eczema. She has even stood up in front of the whole class to explain the research she’s taking part in and show an example of her medicine.”
Her sisters Summayah, 8, and Ayaana, 11, are volunteers in the PELISTAD trial. They don’t have atopic dermatitis, so don’t receive the medicine. Their involvement helps researchers because they can compare their skin against the skin of children of the same age, who have the condition and who are receiving the study medicine.

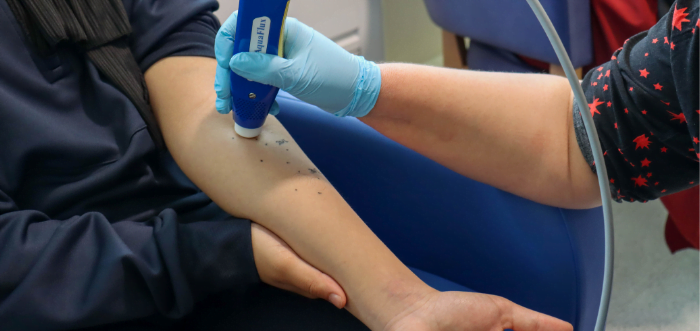
All three sisters come to the Clinical Research Facility for skin examinations as part of the study. These include measuring how well the skin is working as a barrier to water and collecting surface skin samples using sticky-tape discs (pictured right), which are like Sellotape. Infrared light, like that found in television remotes, is also used to look at the structure of the skin and the shape of blood vessels (pictured above).
Ayaana said: “I like taking part in the study because I know I’m helping other children with eczema.”
Mum Safia added: “I can’t thank the staff at Sheffield Children’s enough for what they’ve done for Ammarah. When we started the trial Ammarah had a fear of needles, but they’ve even helped her to overcome that, so she is now happy for me to give her the injections at home.”
Professor Mike Cork, Consultant Dermatologist at Sheffield Children’s, said: “Ammarah has been coming to Sheffield Children’s since she was two years old due to her eczema. When the PELISTAD trial opened, the Dermatology team recognised she was potentially eligible. Clinical trials have precise inclusion and exclusion criteria to ensure the safety of participants and integrity of the trial. Only certain patients or volunteers are therefore suitable. The Research team provided the family with more information about the trial and arranged for Ammarah and her parents to come to the Clinical Research Facility to undergo screening assessments and confirm she was eligible. It’s fantastic to hear the positive effect the drug and also participation in research has had for Ammarah and her family.”
Stuart Gormley, Lead Nurse for Research and Innovation said: “Sheffield Children’s has been involved in dupilumab trials in children and young people since 2015. I’m proud we have been able to support these trials and see the difference it has made to young people with moderate to severe eczema. Ammarah and her sisters have been fantastic throughout the trial and shown a real interest and it has been great to see the change in Ammarah. It will be exciting to see the results of this trial as the equipment and new technology will provide information about the skin beyond what we can see.”
To find out more about what it’s like to take part in research trials at Sheffield Children’s, watch the video below and visit the Research webpage.
To find out more about dermatology research in Sheffield visit: https://sheffielddermatologyresearch.com/
By continuing to use the site, you agree to the use of cookies. more information
The cookie settings on this website are set to "allow cookies" to give you the best browsing experience possible. If you continue to use this website without changing your cookie settings or you click "Accept" below then you are consenting to this.