16 March 2022
Meet eleven-year-old Lillie. Over the last few months, she has been participating in a research trial for an investigational medicine with the Clinical Research Facility (CRF) at Sheffield Children’s.
Lillie has a form of Cerebral Palsy (CP) called Dyskinetic CP, which means that some of her movements are involuntary and can be too quick or abrupt. These involuntary movements can make it difficult for Lillie to do some everyday tasks for herself, such as drinking from a straw or getting dressed.
Her mum, Keighly said: “Lillie was just seven days old when she was diagnosed with Dyskinetic CP, and the CP team at Sheffield Children’s have been so helpful over the years. We’re very lucky that we’re from the Sheffield region – as some other areas just wouldn’t have been able to give us the same input and support.”
Dr Santosh Mordekar, Consultant Paediatric Neurologist said: “Each trial has very specific selection criteria that the patient must fulfil to be eligible. Following a thorough screening process though, Lillie started on the trial and it’s been great to see her frequently throughout the trial.’’
 During the study, Lillie has attended a mix of face-to-face and virtual appointments during which Dr. Mordekar monitored her response to the investigational drug.
During the study, Lillie has attended a mix of face-to-face and virtual appointments during which Dr. Mordekar monitored her response to the investigational drug.Keighly said: “We came in every few weeks for tests, which we had agreed to in the informed consent that we signed.”
“The CRF team have been amazing. We’re nominating Julie – one of Lillie’s nurses – for a DAISY (Diseases Attacking the Immune System) Award because during COVID-19 she’s been there all the time. Even when we couldn’t see her in person, she has been on the phone. Lillie needed to come in to the Emergency Department recently, and Julie came down and see us!”
Stuart Gormley, Lead Nurse for Research and Innovation said: ‘’It has been great to see Lilly take part in clinical research! It can often be a big commitment to take part in a clinical trial but the team will always do what they can to work around participants and their families. We look forward to seeing the results of this trial in due course.’’
Keighly added: “We’re very lucky to be involved in this trial. We’re one of so few in this trial so far. We look forward to seeing the results.”
Thank you for sharing your story, Lillie! To find out more about research trials – and how to take part in them – visit the Research webpage. You might also be interested in...
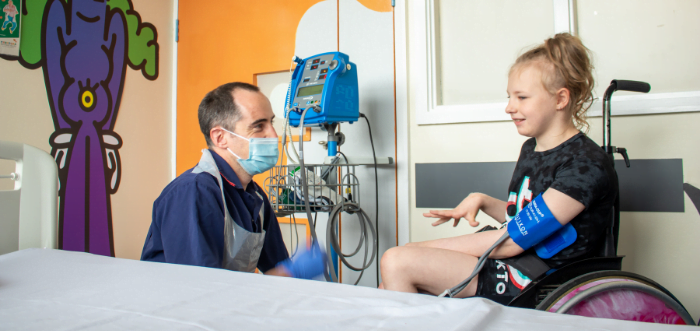
 During the study, Lillie has attended a mix of face-to-face and virtual appointments during which Dr. Mordekar monitored her response to the investigational drug.
During the study, Lillie has attended a mix of face-to-face and virtual appointments during which Dr. Mordekar monitored her response to the investigational drug.